Transcatheter Aortic Valve Replacement TAVR
TAVR is a less invasive, catheter-based technique for replacing the diseased aortic valve. An interventional cardiologist along with a cardiothoracic surgeon will work together in the TAVR procedure. They will guide a new valve into the heart through an incision in the leg while the heart is still beating, using guidance from X-ray and echocardiography.
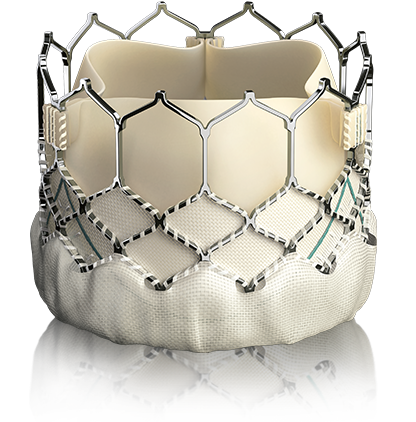
SAPIEN 3 transcatheter heart valve
EDWARDS SAPIEN 3 TRANSCATHETER HEART VALVE The Edwards SAPIEN 3 transcatheter heart valve is a biological tissue valve that will replace your diseased aortic valve. It is available in four sizes, 20, 23, 26 and 29 mm in diameter. Your specialized Heart Team will determine which size is right for you. Edwards’ first transcatheter heart valve was approved commercially in Europe in 2007 and in the United States in 2011. To date, Edwards’ transcatheter heart valves have treated more than 150,000 patients in over 65 countries around the world.
See the clinical difference
Low all-cause mortality and disabling stroke
TAVR with the SAPIEN 3 valve demonstrated 75% lower rates of 30-day all-cause mortality and disabling stroke compared with surgery.
Transformational valve design
Low profile access
Demonstrates significant reduction in major vascular complications
Skirt
Polyethylene terephthalate (PET) outer skirt is designed to minimize paravalvular leak
Valve tissue
Utilizes the same bovine pericardium tissue and processes as Edwards surgical valves
Frame design
Low frame height and open cell geometry promote access to coronary ostia for future interventions
Complete range of valve sizes and access routes expands the treatable patient population
![4.[1]](http://jahangostaresh.com/files/Products/4.[1].png)
How Long Will My Valve Last? How long your tissue valve will last depends on many patient factors and medical conditions. The long-term durability of the SAPIEN 3 valve has not been established. However, regular follow-ups will help your doctor know how your valve is working.
The valve you trust is now available for more patients
The SAPIEN 3 valve is the only valve approved for valve-in-valve procedures due to failure of a surgical aortic or mitral valve.
![3.[1]](http://jahangostaresh.com/files/Products/3.[1].png)







 بروشور راهنمای بیماران (
703 kB)
بروشور راهنمای بیماران (
703 kB)